Gas Type Reference Guide
A
AcetyleneAmmonia
Argon
B
ButaneC
Carbon DioxideCarbon Monoxide
Chlorine
H
HeliumHydrogen
K
KryptonM
MethaneN
NeonNitrogen
Nitrous Oxide
O
OxygenP
PropaneX
XenonAcetylene
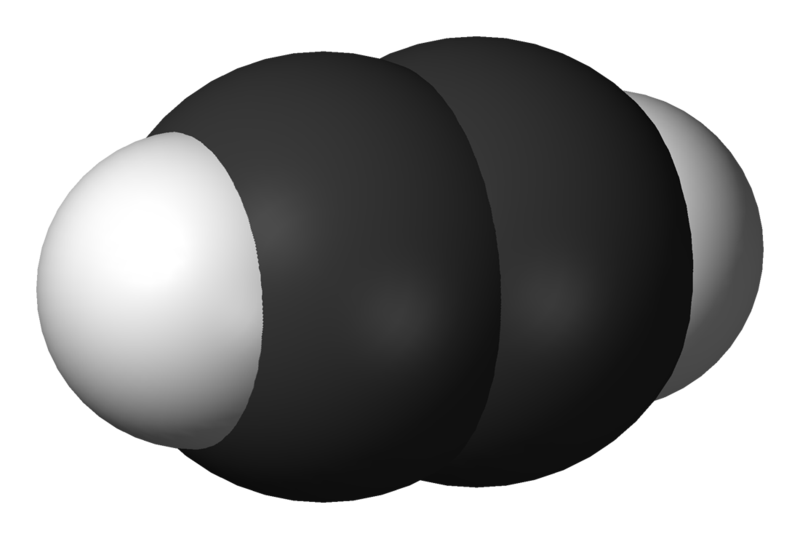 Acetylene (systematic name: ethyne) is the chemical compound with the formula C2H2. It is a hydrocarbon and the simplest alkyne. This colorless gas is widely used as a fuel and a chemical building block. It is unstable in pure form and thus is usually handled as a solution. Pure acetylene is odorless, but commercial grades usually have a marked odor due to impurities.
As an alkyne, acetylene is unsaturated because its two carbon atoms are bonded together in a triple bond. The carbon–carbon triple bond places all four atoms in the same straight line, with CCH bond angles of 180°. Since acetylene is a linear symmetrical molecule, it possesses the D∞h point group.
Acetylene (systematic name: ethyne) is the chemical compound with the formula C2H2. It is a hydrocarbon and the simplest alkyne. This colorless gas is widely used as a fuel and a chemical building block. It is unstable in pure form and thus is usually handled as a solution. Pure acetylene is odorless, but commercial grades usually have a marked odor due to impurities.
As an alkyne, acetylene is unsaturated because its two carbon atoms are bonded together in a triple bond. The carbon–carbon triple bond places all four atoms in the same straight line, with CCH bond angles of 180°. Since acetylene is a linear symmetrical molecule, it possesses the D∞h point group.
| Properties | |
|---|---|
| Molecular formula | C2H2 |
| Molar mass | 26.04 g mol−1 |
| Density | 1.097 g/L = 1.097 kg/m3 |
| Melting point | −80.8 °C; −113.4 °F; 192.3 K (Triple point at 1.27 atm) |
| Boiling point | −84 °C; −119 °F; 189 K (Sublimation point at 1 atm) |
| Solubility in water | slightly soluble |
| Acidity (pKa) | 25[2] |
Ammonia
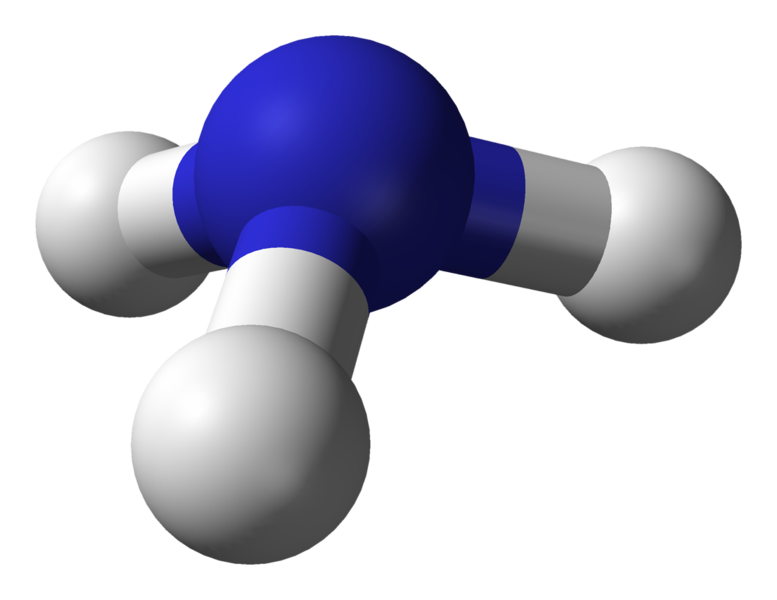 Ammonia or azane is a compound of nitrogen and hydrogen with the formula NH3. It is a colourless gas with a characteristic pungent smell. Ammonia contributes significantly to the nutritional needs of terrestrial organisms by serving as a precursor to food and fertilizers. Ammonia, either directly or indirectly, is also a building-block for the synthesis of many pharmaceuticals and is used in many commercial cleaning products. Although in wide use, ammonia is both caustic and hazardous. The global industrial production of ammonia for 2012 was anticipated to be 198 million tonnes, a 35% increase over the estimated 2006 global output of 146.5 million tonnes.
Ammonia or azane is a compound of nitrogen and hydrogen with the formula NH3. It is a colourless gas with a characteristic pungent smell. Ammonia contributes significantly to the nutritional needs of terrestrial organisms by serving as a precursor to food and fertilizers. Ammonia, either directly or indirectly, is also a building-block for the synthesis of many pharmaceuticals and is used in many commercial cleaning products. Although in wide use, ammonia is both caustic and hazardous. The global industrial production of ammonia for 2012 was anticipated to be 198 million tonnes, a 35% increase over the estimated 2006 global output of 146.5 million tonnes.
NH3 boils at −33.34 °C (−28.012 °F) at a pressure of 1 atmosphere, so the liquid must be stored under high pressure or at low temperature. Household ammonia or ammonium hydroxide is a solution of NH3 in water. The concentration of such solutions is measured in units of the Baumé scale (density), with 26 degrees baumé (about 30% (by weight) ammonia at 15.5 °C) being the typical high-concentration commercial product.
| Properties | |
|---|---|
| Molecular formula | NH3 |
| Molar mass | 17.031 g/mol |
| Appearance | Colourless gas |
| Odor | strong pungent odor |
| Density | 0.86 kg/m3 (1.013 bar at boiling point) 0.73 kg/m3 (1.013 bar at 15 °C) 681.9 kg/m3 at −33.3 °C (liquid)[1] 817 kg/m3 at −80 °C (transparent solid)[2] |
| Melting point | −77.73 °C; −107.91 °F; 195.42 K |
| Boiling point | −33.34 °C; −28.01 °F; 239.81 K |
| Solubility in water | 47% (0 °C) 31% (25 °C) 28% (50 °C)[3] |
| Solubility | soluble in chloroform, ether, ethanol, methanol |
| Vapor pressure | 8573 h Pa |
| Acidity (pKa) | 32.5 (−33 °C),[4] 10.5 (DMSO) |
| Basicity (pKb) | 4.75 |
| Refractive index (nD) | 1.3327 |
| Viscosity | 0.276 cP (-40 °C) |
Argon
 Argon is a chemical element with symbol Ar and atomic number 18. It is in group 18 of the periodic table and is a noble gas. Argon is the third most common gas in the Earth's atmosphere, at 0.93% (9,300 ppm), making it approximately 23.8 times as abundant as the next most common atmospheric gas, carbon dioxide (390 ppm), and more than 500 times as abundant as the next most common noble gas, neon (18 ppm). Nearly all of this argon is radiogenic argon-40 derived from the decay of potassium-40 in the Earth's crust. In the universe, argon-36 is by far the most common argon isotope, being the preferred argon isotope produced by stellar nucleosynthesis in supernovas.
Argon is a chemical element with symbol Ar and atomic number 18. It is in group 18 of the periodic table and is a noble gas. Argon is the third most common gas in the Earth's atmosphere, at 0.93% (9,300 ppm), making it approximately 23.8 times as abundant as the next most common atmospheric gas, carbon dioxide (390 ppm), and more than 500 times as abundant as the next most common noble gas, neon (18 ppm). Nearly all of this argon is radiogenic argon-40 derived from the decay of potassium-40 in the Earth's crust. In the universe, argon-36 is by far the most common argon isotope, being the preferred argon isotope produced by stellar nucleosynthesis in supernovas.
| General properties | |
|---|---|
| Name, symbol, number | argon, Ar, 18 |
| Pronunciation | /ˈɑrɡɒn/ |
| Element category | noble gases |
| Group, period, block | 18 (noble gases), 3, p |
| Standard atomic weight | 39.948(1) |
| Electron configuration | [Ne] 3s2 3p6 2, 8, 8 |
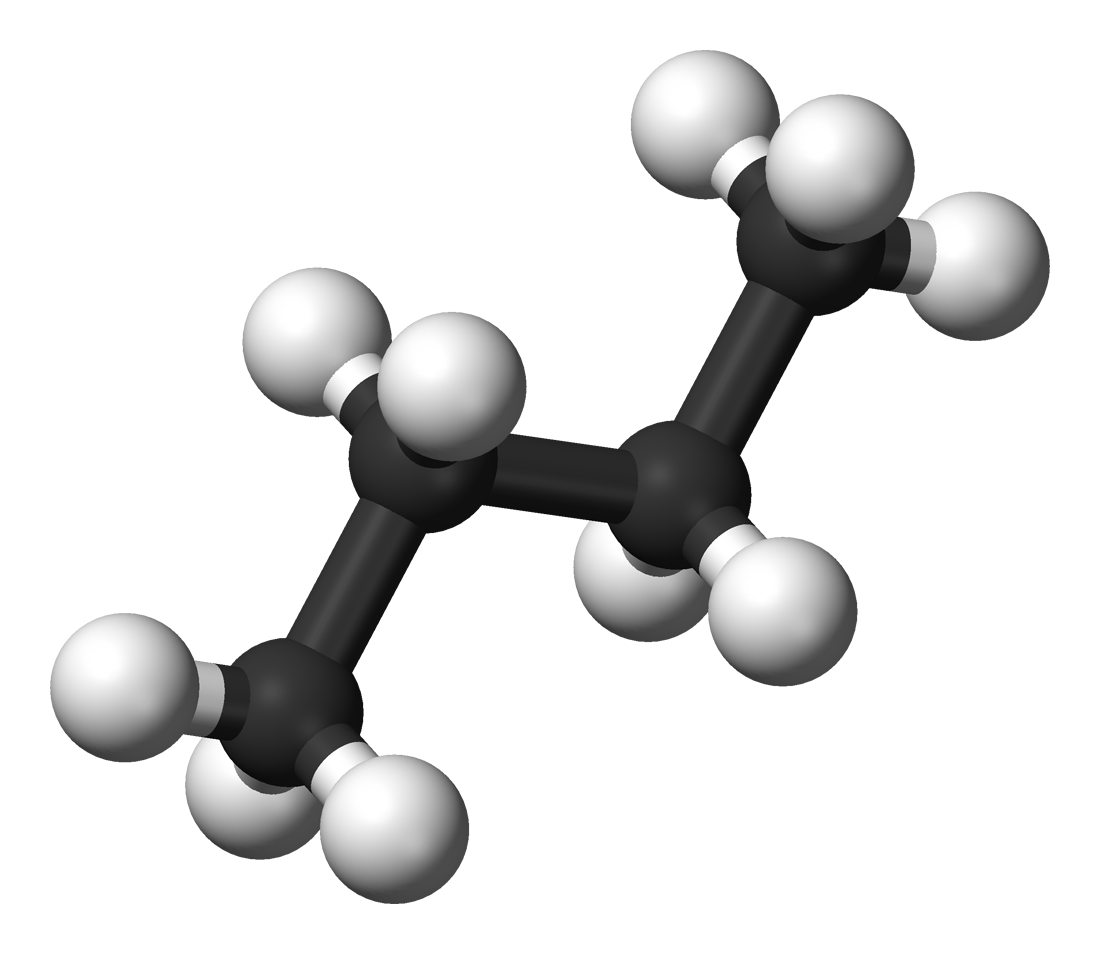
Butane
Carbon Dioxide
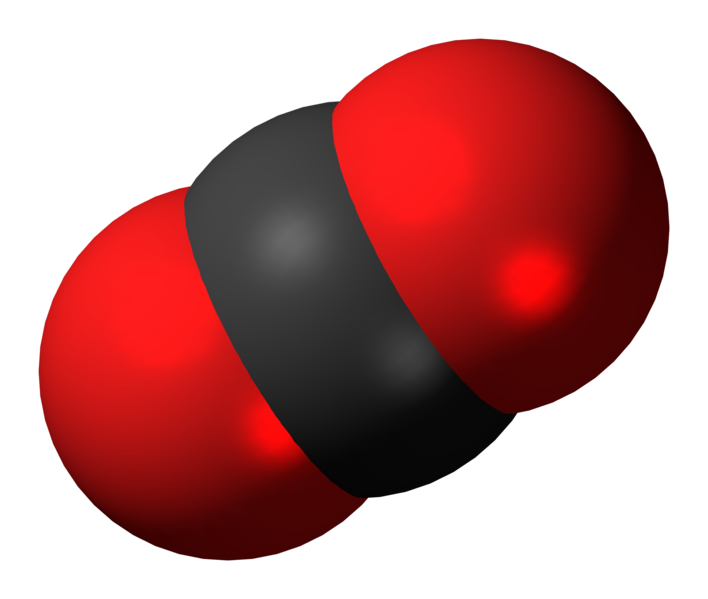 Carbon dioxide (chemical formula CO2) is a naturally occurring chemical compound composed of two oxygen atoms each covalently double bonded to a single carbon atom. It is a gas at standard temperature and pressure and exists in Earth's atmosphere in this state, as a trace gas at a concentration of 0.039 per cent by volume.
Carbon dioxide (chemical formula CO2) is a naturally occurring chemical compound composed of two oxygen atoms each covalently double bonded to a single carbon atom. It is a gas at standard temperature and pressure and exists in Earth's atmosphere in this state, as a trace gas at a concentration of 0.039 per cent by volume.
| Properties | |
|---|---|
| Molecular formula | CO2 |
| Molar mass | 44.01 g mol−1 |
| Appearance | Colorless gas |
| Odor | Odorless |
| Density | 1562 kg/m3 (solid at 1 atm and −78.5 °C) 770 kg/m3 (liquid at 56 atm and 20 °C) 1.977 kg/m3 (gas at 1 atm and 0 °C) |
| Melting point | −78.5 °C; −109.2 °F; 194.7 K (subl.) |
| Boiling point | −56.6 °C; −69.8 °F; 216.6 K (at 5.185 bar) |
| Solubility in water | 1.45 g/L at 25 °C, 100 kPa |
| Vapor pressure | 5.73 MPa (20 °C) |
| Acidity (pKa) | 6.35, 10.33 |
| Refractive index (nD) | 1.1120 |
| Viscosity | 0.07 cP at −78.5 °C |
| Dipole moment | 0 D |
Carbon Monoxide
 Carbon monoxide (CO) is a colorless, odorless, and tasteless gas that is slightly less dense than air. It is toxic to humans and animals when encountered in higher concentrations, although it is also produced in normal animal metabolism in low quantities, and is thought to have some normal biological functions. In the atmosphere, it is spatially variable, short lived, having a role in the formation of ground-level ozone.
Carbon monoxide (CO) is a colorless, odorless, and tasteless gas that is slightly less dense than air. It is toxic to humans and animals when encountered in higher concentrations, although it is also produced in normal animal metabolism in low quantities, and is thought to have some normal biological functions. In the atmosphere, it is spatially variable, short lived, having a role in the formation of ground-level ozone.
Carbon monoxide consists of one carbon atom and one oxygen atom, connected by a triple bond that consists of two covalent bonds as well as one dative covalent bond. It is the simplest oxocarbon, and isoelectronic with the cyanide ion and molecular nitrogen. In coordination complexes the carbon monoxide ligand is called carbonyl.
Carbon monoxide is produced from the partial oxidation of carbon-containing compounds; it forms when there is not enough oxygen to produce carbon dioxide (CO2), such as when operating a stove or an internal combustion engine in an enclosed space. In the presence of oxygen, including atmospheric concentrations, carbon monoxide burns with a blue flame, producing carbon dioxide. Coal gas, which was widely used before the 1960s for domestic lighting, cooking, and heating, had carbon monoxide as a significant fuel constituent. Some processes in modern technology, such as iron smelting, still produce carbon monoxide as a byproduct.
| Properties | |
|---|---|
| Molecular formula | CO |
| Molar mass | 28.010 g/mol |
| Appearance | colorless gas |
| Odor | odorless |
| Density | 789 kg/m3, liquid 1.250 kg/m3 at 0 °C, 1 atm 1.145 kg/m3 at 25 °C, 1 atm |
| Melting point | −205.02 °C; −337.04 °F; 68.13 K |
| Boiling point | −191.5 °C; −312.7 °F; 81.6 K |
| Solubility in water | 27.6 mg/1 L (25 °C) |
| Solubility | soluble in chloroform, acetic acid, ethyl acetate, ethanol, ammonium hydroxide, benzene |
| kH | 1.04 atm-m3/mol |
| Refractive index (nD) | 1.0003364 |
| Dipole moment | 0.122 D |
Chlorine
 Chlorine is a chemical element with symbol Cl and atomic number 17. Chlorine is in the halogen group (17) and is the second lightest halogen after fluorine. The element is a yellow-green gas under standard conditions, where it forms diatomic molecules. It has the highest electron affinity and the third highest electronegativity of all the reactive elements; for this reason, chlorine is a strong oxidizing agent. Free chlorine is rare on Earth, and is usually a result of direct or indirect oxidation by oxygen.
Chlorine is a chemical element with symbol Cl and atomic number 17. Chlorine is in the halogen group (17) and is the second lightest halogen after fluorine. The element is a yellow-green gas under standard conditions, where it forms diatomic molecules. It has the highest electron affinity and the third highest electronegativity of all the reactive elements; for this reason, chlorine is a strong oxidizing agent. Free chlorine is rare on Earth, and is usually a result of direct or indirect oxidation by oxygen.
| General properties | |
|---|---|
| Name, symbol, number | chlorine, Cl, 17 |
| Pronunciation | |
| Element category | diatomic nonmetal |
| Group, period, block | 17 (halogens), 3, p |
| Standard atomic weight | 35.45(1) |
| Electron configuration | [Ne] 3s2 3p5 2, 8, 7 |
Helium
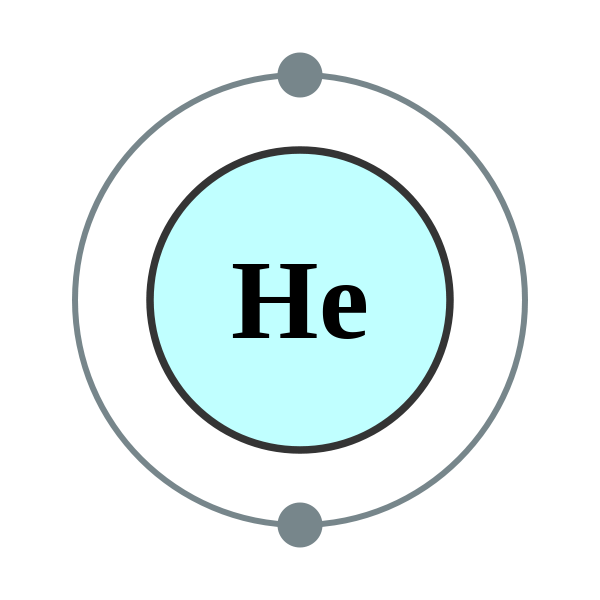 Helium is a chemical element with symbol He and atomic number 2. It is a colorless, odorless, tasteless, non-toxic, inert, monatomic gas that heads the noble gas group in the periodic table. Its boiling and melting points are the lowest among the elements and it exists only as a gas except in extreme conditions.
Helium is a chemical element with symbol He and atomic number 2. It is a colorless, odorless, tasteless, non-toxic, inert, monatomic gas that heads the noble gas group in the periodic table. Its boiling and melting points are the lowest among the elements and it exists only as a gas except in extreme conditions.
| General properties | |
|---|---|
| Name, symbol, number | helium, He, 2 |
| Element category | noble gases |
| Group, period, block | 18 (noble gases), 1, s |
| Standard atomic weight | 4.002602(2) |
| Electron configuration | 1s2 2 |
Hydrogen

| General properties | |
|---|---|
| Name, symbol, number | hydrogen, H, 1 |
| Element category | diatomic nonmetal |
| Group, period, block | 1, 1, s |
| Standard atomic weight | 1.008(1) |
| Electron configuration | 1s1 1 |
Krypton
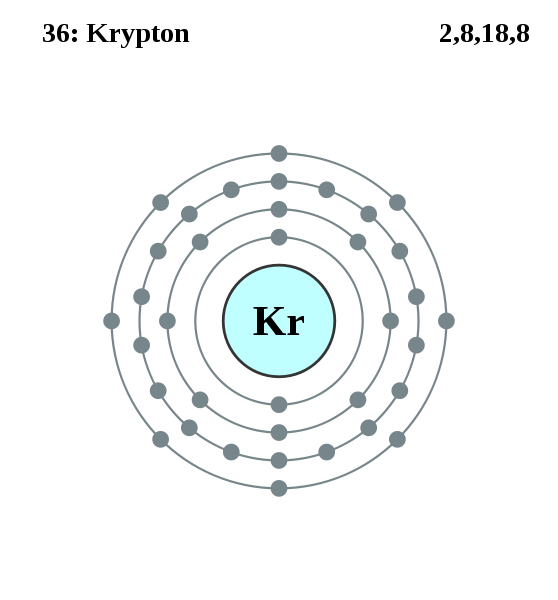 Krypton (from Greek: κρυπτός kryptos "the hidden one") is a chemical element with symbol Kr and atomic number 36. It is a member of group 18 (noble gases) elements. A colorless, odorless, tasteless noble gas, krypton occurs in trace amounts in the atmosphere, is isolated by fractionally distilling liquified air, and is often used with other rare gases in fluorescent lamps. Krypton is inert for most practical purposes.
Krypton (from Greek: κρυπτός kryptos "the hidden one") is a chemical element with symbol Kr and atomic number 36. It is a member of group 18 (noble gases) elements. A colorless, odorless, tasteless noble gas, krypton occurs in trace amounts in the atmosphere, is isolated by fractionally distilling liquified air, and is often used with other rare gases in fluorescent lamps. Krypton is inert for most practical purposes.
| General properties | |
|---|---|
| Name, symbol, number | krypton, Kr, 36 |
| Element category | noble gases |
| Group, period, block | 18 (noble gases), 4, p |
| Standard atomic weight | 83.798 |
| Electron configuration | [Ar] 3d10 4s2 4p6 2, 8, 18, 8 |
| History | |
| Discovery | William Ramsay and Morris Travers(1898) |
| First isolation | William Ramsay and Morris Travers (1898) |
Methane
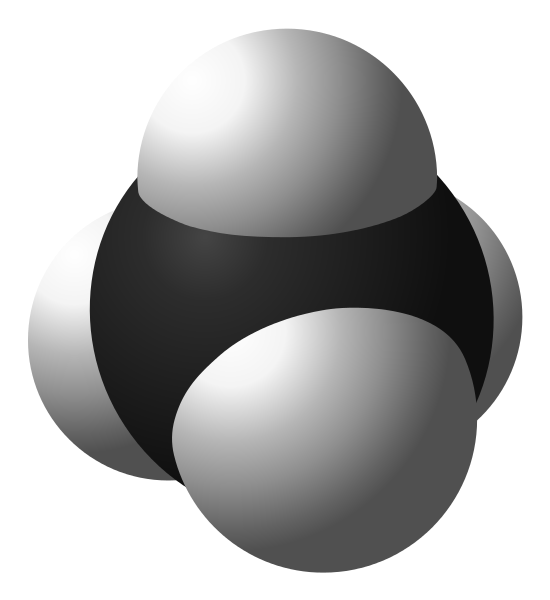 Methane (/ˈmɛθeɪn/, /ˈmiːθeɪn/ or /ˈmɛtæn/) is a chemical compound with the chemical formula CH
4 (one atom of carbon and four atoms of hydrogen). It is the simplest alkane and the main component of natural gas. The relative abundance of methane makes it an attractive fuel. However, because it is a gas at normal conditions, methane is difficult to transport from its source.
Methane (/ˈmɛθeɪn/, /ˈmiːθeɪn/ or /ˈmɛtæn/) is a chemical compound with the chemical formula CH
4 (one atom of carbon and four atoms of hydrogen). It is the simplest alkane and the main component of natural gas. The relative abundance of methane makes it an attractive fuel. However, because it is a gas at normal conditions, methane is difficult to transport from its source.
| Properties | |
|---|---|
| Molecular formula | CH4 |
| Molar mass | 16.04 g mol−1 |
| Appearance | Colorless gas |
| Odor | Odorless |
| Density | 0.656g/L @ 25°C, 1 atm 0.716g/L @ 0°C, 1 atm 0.42262 g cm−3 (at 111 K)[2] |
| Melting point | −182.5 °C; −296.4 °F; 90.7 K |
| Boiling point | −164 to −160 °C; −263 to −256 °F; 109 to 113 K |
| Solubility in water | 22.7 mg L−1 |
| Solubility | soluble in ethanol, diethyl ether, benzene, toluene, methanol, acetone |
| log P | 1.09 |
| kH | 14 nmol Pa−1 kg−1 |
| Structure | |
| Molecular shape | Tetrahedron |
| Dipole moment | 0 D |
Neon
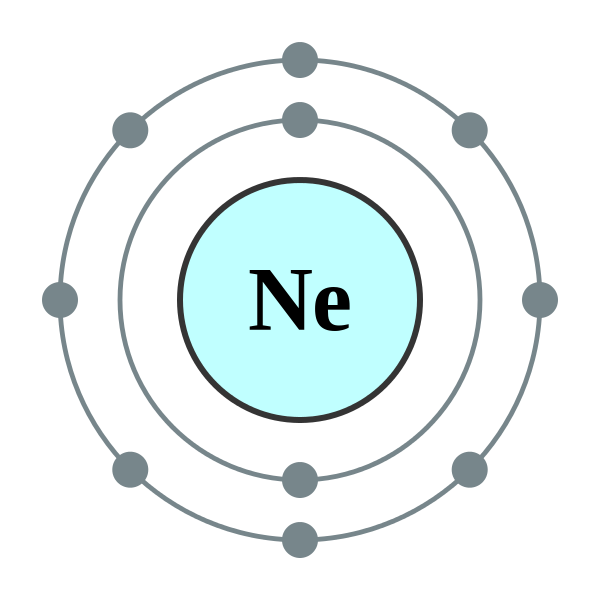 Neon is a chemical element with symbol Ne and atomic number 10. It is in group 18 (noble gases) of the periodic table. Neon is a colorless, odorless, inert monatomic gas under standard conditions, with about two-thirds the density of air. It was discovered (along with krypton and xenon) in 1898 as one of the three residual rare inert elements remaining in dry air, after nitrogen, oxygen, argon and carbon dioxide are removed. Neon was the second of these three rare gases to be discovered, and was immediately recognized as a new element from its bright red emission spectrum. The name neon is derived from the Greek word, νέον, neuter singular form of νέος [neos], meaning new. Neon is chemically inert and forms no uncharged chemical compounds.
Neon is a chemical element with symbol Ne and atomic number 10. It is in group 18 (noble gases) of the periodic table. Neon is a colorless, odorless, inert monatomic gas under standard conditions, with about two-thirds the density of air. It was discovered (along with krypton and xenon) in 1898 as one of the three residual rare inert elements remaining in dry air, after nitrogen, oxygen, argon and carbon dioxide are removed. Neon was the second of these three rare gases to be discovered, and was immediately recognized as a new element from its bright red emission spectrum. The name neon is derived from the Greek word, νέον, neuter singular form of νέος [neos], meaning new. Neon is chemically inert and forms no uncharged chemical compounds.
| General properties | |
|---|---|
| Name, symbol, number | neon, Ne, 10 |
| Element category | noble gases |
| Group, period, block | 18 (noble gases), 2, p |
| Standard atomic weight | 20.1797(6) |
| Electron configuration | [He] 2s2 2p6 2, 8 |
Nitrogen
 Nitrogen, symbol N, is the chemical element of atomic number 7. At room temperature, it is a gas of diatomic molecules and is colorless and odorless. Nitrogen is a common element in the universe, estimated at about seventh in total abundance in our galaxy and the Solar System. On Earth, the element is primarily found as the free element; it forms about 80% of the Earth's atmosphere. The element nitrogen was discovered as a separable component of air by Scottish physician Daniel Rutherford in 1772.
Nitrogen, symbol N, is the chemical element of atomic number 7. At room temperature, it is a gas of diatomic molecules and is colorless and odorless. Nitrogen is a common element in the universe, estimated at about seventh in total abundance in our galaxy and the Solar System. On Earth, the element is primarily found as the free element; it forms about 80% of the Earth's atmosphere. The element nitrogen was discovered as a separable component of air by Scottish physician Daniel Rutherford in 1772.
| General properties | |
|---|---|
| Name, symbol, number | nitrogen, N, 7 |
| Element category | diatomic nonmetal |
| Group, period, block | 15 (pnictogens), 2, p |
| Standard atomic weight | 14.007(1) |
| Electron configuration | [He] 2s2 2p3 2, 5 |
Nitrous oxide
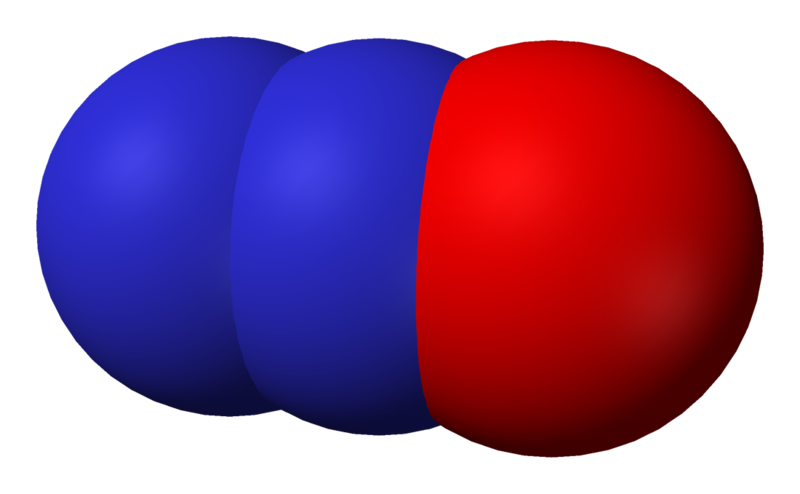 Nitrous oxide, commonly known as laughing gas, nitrous, nitro, or NOS[1] is a chemical compound with the formula N
2O. It is an oxide of nitrogen. At room temperature, it is a colourless, non-flammable gas, with a slightly sweet odour and taste. It is used in surgery and dentistry for its anaesthetic and analgesic effects. It is known as "laughing gas" due to the euphoric effects of inhaling it, a property that has led to its recreational use as a dissociative anaesthetic. It is also used as an oxidizer in rocketry and in motor racing to increase the power output of engines. At elevated temperatures, nitrous oxide is a powerful oxidizer similar to molecular oxygen
Nitrous oxide, commonly known as laughing gas, nitrous, nitro, or NOS[1] is a chemical compound with the formula N
2O. It is an oxide of nitrogen. At room temperature, it is a colourless, non-flammable gas, with a slightly sweet odour and taste. It is used in surgery and dentistry for its anaesthetic and analgesic effects. It is known as "laughing gas" due to the euphoric effects of inhaling it, a property that has led to its recreational use as a dissociative anaesthetic. It is also used as an oxidizer in rocketry and in motor racing to increase the power output of engines. At elevated temperatures, nitrous oxide is a powerful oxidizer similar to molecular oxygen
| Properties | |
|---|---|
| Molar mass | 44.013 g/mol |
| Appearance | colourless gas |
| Density | 1.977 g/L (gas) |
| Melting point | −90.86 °C (182.29 K) |
| Boiling point | −88.48 °C (184.67 K) |
| Solubility in water | 0.15 g/100 ml (15 °C) |
| Solubility | soluble in alcohol, ether, sulfuric acid |
| log P | 0.35 |
| Vapor pressure | 5150 kPa (20 °C) |
| Refractive index (nD) | 1.330 |
Oxygen
 Oxygen is a chemical element with symbol O and atomic number 8. It is a member of the chalcogen group on the periodic table and is a highly reactive nonmetallic element and oxidizing agent that readily forms compounds (notably oxides) with most elements.[1] By mass, oxygen is the third-most abundant element in the universe, after hydrogen and helium.[2] At STP, two atoms of the element bind to form dioxygen, a diatomic gas that is colorless, odorless, and tasteless; with the formula O
2.
Oxygen is a chemical element with symbol O and atomic number 8. It is a member of the chalcogen group on the periodic table and is a highly reactive nonmetallic element and oxidizing agent that readily forms compounds (notably oxides) with most elements.[1] By mass, oxygen is the third-most abundant element in the universe, after hydrogen and helium.[2] At STP, two atoms of the element bind to form dioxygen, a diatomic gas that is colorless, odorless, and tasteless; with the formula O
2.
| General properties | |
|---|---|
| Name, symbol, number | oxygen, O, 8 |
| Element category | diatomic nonmetal, chalcogen |
| Group, period, block | 16 (chalcogens), 2, p |
| Standard atomic weight | 15.999(4) |
| Electron configuration | [He] 2s2 2p4 2, 6 |
Propane
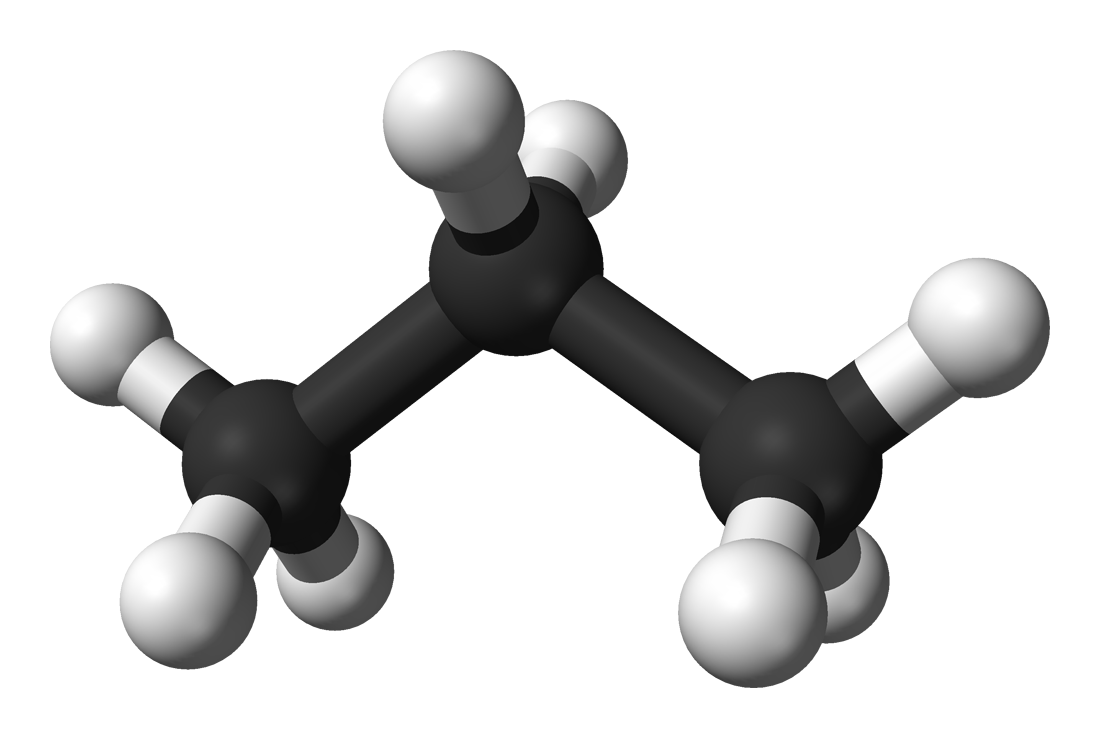 Propane is a three-carbon alkane with the molecular formula C 3H 8, normally a gas, but compressible to a transportable liquid. A by-product of natural gas processing and petroleum refining, it is commonly used as a fuel for engines, oxy-gas torches, barbecues, portable stoves, and residential central heating. Propane is one of a group of liquefied petroleum gases. The others include butane, propylene, butadiene, butylene, isobutylene and mixtures thereof.
Propane is a three-carbon alkane with the molecular formula C 3H 8, normally a gas, but compressible to a transportable liquid. A by-product of natural gas processing and petroleum refining, it is commonly used as a fuel for engines, oxy-gas torches, barbecues, portable stoves, and residential central heating. Propane is one of a group of liquefied petroleum gases. The others include butane, propylene, butadiene, butylene, isobutylene and mixtures thereof.
| Properties[2] | |
|---|---|
| Molecular formula | C3H8 |
| Molar mass | 44.10 g mol−1 |
| Appearance | Colorless gas |
| Odor | Odorless |
| Density | 2.0098 mg mL−1 (at 0 °C, 101.3 kPa) |
| Melting point | −187.7 °C; −305.8 °F; 85.5 K |
| Boiling point | −42.2 to −42.04 °C; −44.1 to −43.67 °F; 230.9 to 231.11 K |
| Solubility in water | 40 mg L−1 (at 0 °C) |
| log P | 2.236 |
| Vapor pressure | 853.16 kPa (at 21.1 °C) |
| kH | 15 nmol Pa−1 kg−1 |
Xenon
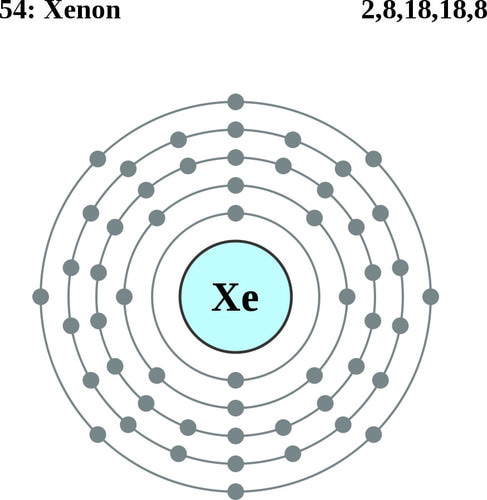 Xenon is a chemical element with the symbol Xe and atomic number 54. It is a colorless, heavy, odorless noble gas, that occurs in the Earth's atmosphere in trace amounts.[8] Although generally unreactive, xenon can undergo a few chemical reactions such as the formation of xenon hexafluoroplatinate, the first noble gas compound to be synthesized.[9][10][11]
Naturally occurring xenon consists of eight stable isotopes. There are also over 40 unstable isotopes that undergo radioactive decay. The isotope ratios of xenon are an important tool for studying the early history of the Solar System.[12] Radioactive xenon-135 is produced from iodine-135 as a result of nuclear fission, and it acts as the most significant neutron absorber in nuclear reactors.[13]
Xenon is a chemical element with the symbol Xe and atomic number 54. It is a colorless, heavy, odorless noble gas, that occurs in the Earth's atmosphere in trace amounts.[8] Although generally unreactive, xenon can undergo a few chemical reactions such as the formation of xenon hexafluoroplatinate, the first noble gas compound to be synthesized.[9][10][11]
Naturally occurring xenon consists of eight stable isotopes. There are also over 40 unstable isotopes that undergo radioactive decay. The isotope ratios of xenon are an important tool for studying the early history of the Solar System.[12] Radioactive xenon-135 is produced from iodine-135 as a result of nuclear fission, and it acts as the most significant neutron absorber in nuclear reactors.[13]
| General properties | |
|---|---|
| Name, symbol, number | xenon, Xe, 54 |
| Element category | noble gases |
| Group, period, block | 18 (noble gases), 5, p |
| Standard atomic weight | 131.293(6) |
| Electron configuration | [Kr] 5s2 4d10 5p6 2, 8, 18, 18, 8 |

